Introduction
Every living cell depends on water. Water carries nutrients, removes waste, and maintains the internal environment. Scientists often use simple models to understand how water behaves in and around cells. One powerful learning tool is a simcell with a water-permeable membrane.
Researchers designed this artificial system to mimic the behavior of a real biological cell. It allows students and scientists to observe how water moves when the environment changes. This model helps explain one of the most important biological processes: osmosis.
When you study a semicell with a water-permeable membrane, you see how concentration differences drive water movement. The model shows why cells swell in some environments and shrink in others. Because the concept appears simple, many people underestimate its importance. Yet this basic idea explains many processes in biology, medicine, and environmental science.
This article explains how a simcell with a water-permeable membrane works, why scientists use it, and how it helps us understand real cells. The explanation uses clear language so anyone can easily understand the concept.
Understanding the Concept of a SimCell
A SimCell, or simulated cell, represents a simplified version of a real biological cell. Scientists create it to study how membranes control the movement of molecules.
A typical SimCell contains water and dissolved substances such as sugars or salts. A thin membrane surrounds the internal solution. This membrane controls which molecules can pass through it.
In many experiments, researchers design a simcell with a water-permeable membrane. The membrane allows water molecules to pass freely but blocks larger solute molecules. This selective behavior creates an environment where water movement becomes easy to observe.
This design helps students visualize how a real cell behaves when placed in different solutions. The SimCell acts like a miniature laboratory where scientists can test biological ideas without using living cells.
Because the model stays simple, learners can focus on the core principle: water always moves toward higher solute concentration.
What Makes a Water Permeable Membrane Special
A membrane controls the movement of substances between two environments. In biological systems, the cell membrane regulates what enters and leaves the cell.
In a semicell with a water-permeable membrane, water molecules can pass through easily. However, the membrane prevents most dissolved substances from crossing.
This property creates a difference between the inside and outside environments. Water molecules respond to this difference and move across the membrane.
The movement of water depends on concentration gradients. If the outside environment contains more water molecules, water flows into the SimCell. If the inside environment contains more water, water flows outward.
Because of this selective permeability, the SimCell demonstrates how real cells maintain balance.
Osmosis: The Driving Force Behind Water Movement
Water movement across a membrane occurs by osmosis. Osmosis describes the natural process in which water travels across a semipermeable membrane toward a higher solute concentration.
When scientists use a semicell with a water-permeable membrane, they observe osmosis clearly. The internal solution often contains dissolved molecules that cannot leave the cell. These molecules attract water.
Water molecules move from the region with more free water toward the region with less free water. This movement continues until the concentration difference decreases.
Osmosis explains many biological processes. It controls how plant roots absorb water, how cells maintain structure, and how the body regulates fluid balance.
The SimCell model allows students to observe these effects simply and visually.
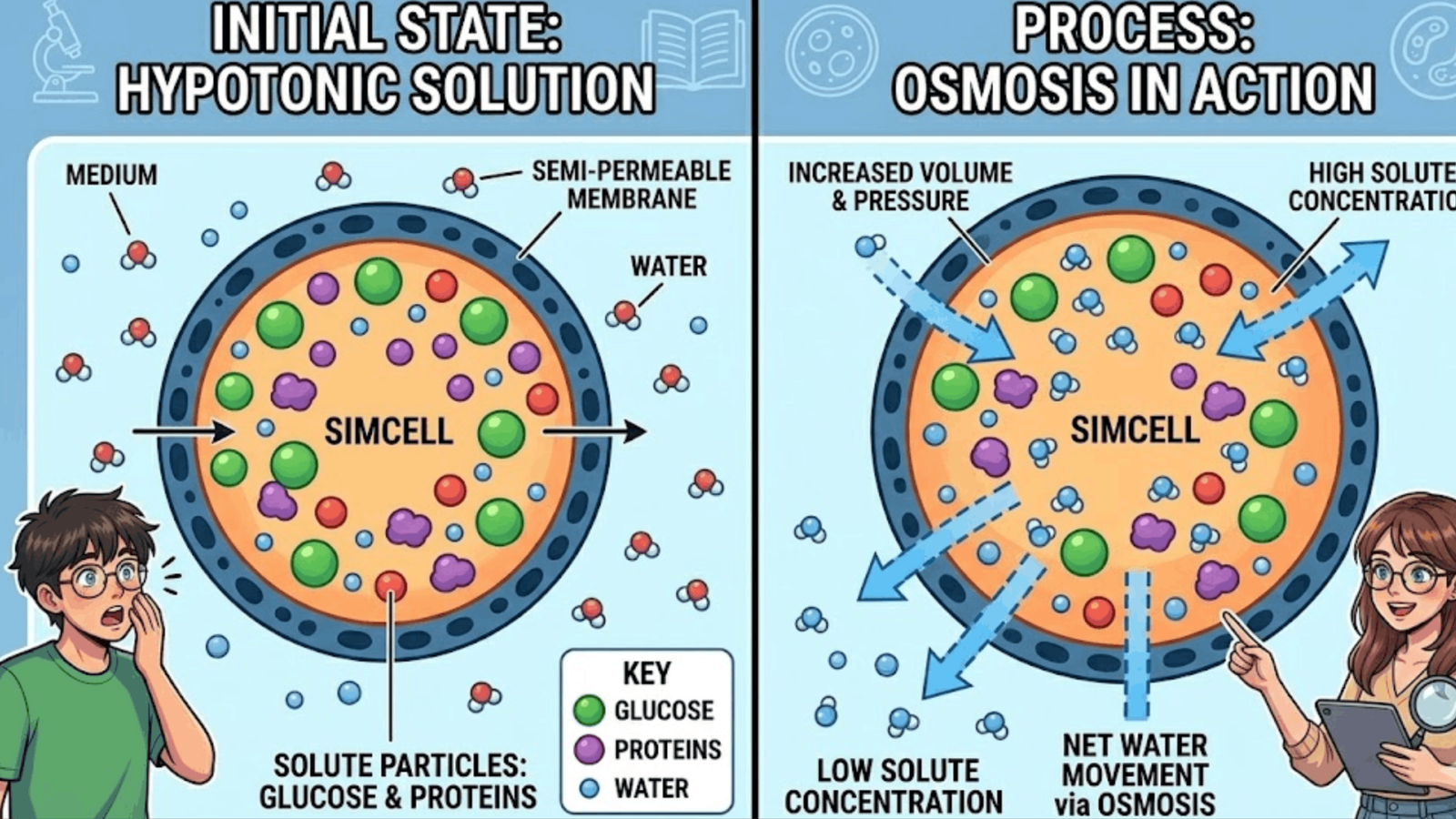
Hypotonic Environments and Cell Expansion
A hypotonic environment has a lower solute concentration than the intracellular environment.
When scientists place a cell with a water-permeable membrane in a hypotonic solution, the outside environment contains more water molecules. Water moves into the SimCell through osmosis.
As water enters, the internal volume increases. The SimCell can expand and even burst if the membrane stretches too much.
This process mirrors what happens in real biological cells. Animal cells can swell when they absorb too much water. Plant cells also take in water, but their strong cell walls prevent bursting.
The SimCell demonstrates how delicate the water balance becomes in biological systems.
Hypertonic Environments and Cell Shrinkage
A hypertonic environment contains a higher concentration of solutes compared to the inside of the cell.
When researchers place a SimCell with a water-permeable membrane in such an environment, water moves out of the SimCell.
The external solution attracts water because it contains more solute particles. As water leaves the SimCell, the internal volume decreases.
The SimCell shrinks and loses mass.
This effect also occurs in real cells. Red blood cells shrink when they enter highly concentrated solutions. Plants lose turgor pressure when they lose water, causing wilting.
The SimCell model allows students to observe this process clearly.
Isotonic Conditions and Balanced Water Movement
An isotonic environment contains the same solute concentration as the internal environment of the cell.
When scientists place a semicell with a water-permeable membrane in an isotonic solution, water moves in both directions across the membrane.
Water molecules continuously enter and exit the cell. However, the rates remain equal. Because the flow balances itself, the SimCell maintains a stable size.
This situation represents the ideal condition for many animal cells in the body. Human cells function best when the surrounding fluid remains isotonic.
The SimCell model helps learners understand why the body carefully regulates salt and water balance.
Why Scientists Use SimCell Models
Researchers and educators rely on simple models to explain complex biological processes. A semicell with a water-permeable membrane provides a clear visual example of membrane transport.
The model helps students understand how concentration gradients influence water movement. It removes unnecessary complexity so learners can focus on the main concept.
Scientists also use SimCell experiments to test theoretical predictions about diffusion and osmotic pressure.
Because the system remains controlled, researchers can change one variable at a time. They can alter solute concentration, membrane permeability, or temperature to observe different results.
This flexibility makes the SimCell a powerful teaching and research tool.
Real-Life Applications of Osmosis
The principles demonstrated by a simcell with a water-permeable membrane appear in many real-life situations.
In medicine, doctors use osmotic principles when they deliver intravenous fluids. The fluid must match the body’s osmotic balance to prevent cell damage.
In agriculture, farmers manage soil water balance to protect crops. Plants use osmotic pressure to pull water into their roots.
Food preservation also uses osmotic effects. High concentrations of salt or sugar draw water out of bacteria, preventing growth.
These examples show how a simple biological concept affects everyday life.
Understanding Solute Concentration
Solute concentration determines the rate of water movement across a membrane. Solutes include substances such as salts, sugars, and proteins.
In a simcell with a water-permeable membrane, the internal solution may contain molecules such as glucose or dextrose. These molecules remain trapped inside the membrane.
Because they cannot escape, they create an osmotic pull that attracts water molecules.
The greater the solute concentration inside the SimCell, the stronger the pull for water. This relationship explains why cells swell or shrink in response to their surrounding environment.
Understanding concentration gradients helps scientists predict the direction of water movement.
The Educational Power of SimCell Experiments
Teachers often introduce a cell with a water-permeable membrane in classrooms to demonstrate osmosis visually.
Students can measure the mass of the SimCell before and after placing it in different solutions. They observe changes in weight and volume as water moves across the membrane.
This hands-on approach improves learning because students witness the process directly.
Experiments like these strengthen scientific thinking. Students learn how to form hypotheses, analyze results, and interpret data.
The SimCell model transforms abstract theory into visible evidence.
Membrane Selectivity and Cellular Survival
Every living cell relies on selective permeability. The cell membrane must allow certain substances to pass while blocking others.
In a semicell with a water-permeable membrane, the membrane allows water to move but restricts larger solute molecules.
This selectivity creates controlled movement across the boundary.
Real cells use similar strategies. They contain specialized proteins that regulate the transport of ions, nutrients, and waste.
Without this control system, cells could not maintain internal stability.
The SimCell model demonstrates how crucial membrane selectivity becomes for survival.
The Role of Water in Cellular Life
Water supports nearly every biological process. Water dissolves substances, carries nutrients, and helps control temperature.
When scientists examine a simcell with a water-permeable membrane, they observe how water maintains balance inside a cell-like structure.
Water movement ensures that nutrients remain available and waste products leave the cell efficiently.
If water balance is disrupted, cells quickly lose function.
This fact explains why organisms must carefully regulate hydration.
The SimCell experiment highlights the importance of water in maintaining life.
Common Misunderstandings About Osmosis
Many people misunderstand osmosis. Some assume water always moves into cells. Others believe solutes move through the membrane easily.
However, a simcell with a water-permeable membrane demonstrates the real mechanism.
Water movement depends entirely on concentration differences. The membrane determines which molecules can pass.
If the membrane blocks solutes, only water responds to the gradient.
Understanding this concept prevents confusion in biology studies.
Why SimCell Models Matter in Modern Science
Modern biology relies on advanced tools and technology. Yet simple models still play an important role in scientific education.
A simcell with a water-permeable membrane represents one of the most effective models for explaining membrane transport.
The model builds a strong foundation for understanding more complex topics such as cell signaling, metabolism, and physiology.
Students who understand osmotic principles can easily grasp advanced concepts later.
Because of this educational value, SimCell experiments continue to appear in classrooms worldwide.
Conclusion
Science often begins with simple ideas that reveal deep truths. A simcell with a water-permeable membrane offers one of the clearest demonstrations of how cells control water movement.
The model shows how osmosis drives water across membranes in response to concentration differences. Depending on the environment, the SimCell can swell, shrink, or remain stable.
Through this simple experiment, students and researchers gain insight into essential biological processes. The concept explains how cells maintain balance, survive in different environments, and support life itself.
Understanding this model strengthens knowledge of biology and highlights the incredible design of living systems.
For more insights, you may also like to read about: Laaster







Leave a Reply